A practical guide to the different types of chemical recycling and their applications.
This guide outlines the primary chemical recycling methods, the types of plastics they target, key terminology, and useful ways to categorize and compare them. Whether you're reviewing for work, research, education, or general interest, it serves as a streamlined reference for understanding the fundamentals of advanced plastic waste recycling.
As advanced plastic recycling technologies – including chemical recycling – continue to evolve, they’re becoming increasingly relevant as a complimentary solution to mechanical recycling—not only for engineers and policymakers, but also for professionals in sustainability, packaging, education, and materials science. New technologies are emerging, terminology is evolving, and regulatory frameworks are shaping how these innovations develop and scale as part of broader efforts toward sustainable plastic solutions.
Rather than simply listing recycling types, this guide compares them across several dimensions: the plastics they can process, the outputs they produce, their energy requirements, and their current stage of adoption. It also highlights where newer approaches like Hydrochemolytic™ Technology developed by Aduro Clean Technologies fit in—and how they differ from more established or conventional methods aiming to address plastic pollution, and scale plastic waste recycling.
Why chemical recycling matters for plastic waste management and sustainable plastic solutions: the role of extended producer responsibility (EPR) in promoting advanced plastic recycling
As recycling technologies evolve, so do the policies shaping their adoption. Extended Producer Responsibility (EPR) frameworks are becoming more common around the world, shifting the responsibility for plastic waste away from taxpayers and toward the companies that produce it.
Under EPR, producers are incentivized to support or invest in better recycling solutions, including advanced plastic recycling technologies – such as chemical recycling – that go beyond traditional mechanical methods. These policies help drive innovation, promote circularity, and influence what kinds of plastic waste recycling systems are viable at scale.
That’s why it’s worth understanding the different types of chemical recycling—how they work, what plastics they’re compatible with, and how they compare in terms of outputs, energy use, commercial potential, long-term impact, and real-world applications. Together, these efforts contribute to building sustainable plastic solutions that reduce pollution and maximize resource recovery.
Types of chemical recycling technologies for plastic waste recycling
Here’s a quick overview of the diverse types of chemical recycling before we dive deeper into each of them. With so many methods out there, it can feel overwhelming—especially if it’s been a while since your last chemistry class. Don’t worry though, we’ll break it down (pun definitely intended).
-
Pyrolysis: Heats plastic without oxygen to break it into oils and gases.
-
Gasification: Uses limited oxygen at high temperatures to produce syngas.
-
Depolymerization: Breaks plastics down into monomers using heat or catalysts.
-
Solvolysis: Uses solvents to chemically break plastic bonds (e.g., glycolysis).
-
Hydrogenolysis: Uses hydrogen and catalysts to cleave polymer chains.
-
Hydrocracking: Combines hydrogen and catalytic cracking to produce fuels.
-
Enzymolysis / Enzymatic Recycling: Method using engineered enzymes to digest plastic polymers.
-
Dissolution / Solvent-Based Purification: Dissolves plastic to remove additives without breaking the polymer.
-
Aduro’s Hydrochemolytic™ Technology: Uses water, low temperatures, and unique catalysts to break down polymers into reusable components.
GlossaryChemical recycling terms simplifiedAlkanes: Simple hydrocarbon molecules made of only carbon and hydrogen, often found in fuels like propane and butane. Alkanes can be recovered from plastics and used as energy sources or raw materials. Catalyst: Substance that speeds up a chemical reaction without being consumed. In recycling, catalysts help break down polymers into monomers more efficiently. Cleave: To split or break a chemical bond between atoms in a molecule. Cleaving is how some recycling methods break long plastic chains into smaller, reusable parts. Depolymerization: The process of breaking down a polymer into monomers. Enzymes: Natural proteins that act as biological catalysts. Some experimental recycling methods use enzymes to break down plastics, mimicking natural degradation processes. Feedstock: A raw material used to make other products through chemical processes. In recycling, broken-down plastic components can become feedstocks for producing new plastics, fuels, or chemicals. Monomer: Basic unit, building block or single “link” in a polymer chain Oxidize: To chemically react with oxygen, often resulting in the loss of electrons. In recycling, oxidation can help break down plastics or convert them into gases or oils — typically using just enough oxygen to trigger a chemical reaction, but not enough to cause combustion or burning. Polymer: Long chain of repeating molecules or monomers. Re-precipitate: To make a dissolved material solid again, usually by changing the temperature or adding another chemical. In recycling, this helps recover purified plastic after it's been dissolved to remove additives. Solvent: A liquid that dissolves another substance. Syngas: A mix of hydrogen (H₂) and carbon monoxide (CO) gas, used for fuel or chemicals. |
How to categorize chemical recycling types for plastic waste recycling
Effective Ways to Categorize Plastic Waste Recycling Methods
Because plastic recycling technologies vary widely, experts organize them into groups based on key factors like chemical process, energy use, and material compatibility. These classifications help make sense of complex methods and reveal their strengths and challenges. Here, we introduce the main ways to categorize recycling types, providing a foundation for deeper understanding.
Categorizing Chemical Recycling Methods by Compatible Plastic Types: How Plastic Waste Recycling Methods Vary by Plastic Compatibility
One way to classify how plastics are recycled, is to look at what types of plastics the recycling method is used on..
Type of plastic |
Recyclable via… |
Produces |
|
|
|
Polyethylene Terephthalate (PET, PETE) |
Depolymerization |
Monomers (e.g., BHET or TPA and EG) |
|
Solvolysis |
|||
|
|
High-Density Polyethylene (HDPE) |
Pyrolysis |
Oils, waxes, or synthetic fuels |
|
Gasification |
Syngas |
||
|
Hydrochemolytic™ Oil |
|||
|
|
Polyvinyl Chloride (PVC) |
Rarely recycled chemically |
Chemical recycling is uncommon due to toxic chlorine content |
 |
Low-Density Polyethylene (LDPE) Plastic bags, squeeze bottles, cling film |
Pyrolysis |
Oils, waxes, or synthetic fuels |
|
Gasification |
Syngas |
||
|
Hydrochemolytic™ Oil |
|||
|
|
Polypropylene (PP) |
Pyrolysis |
Fuels or waxes |
|
Hydrogenolysis |
Liquid alkanes |
||
|
Hydrochemolytic™ Oil |
|||
|
|
Polystyrene (PS) |
Depolymerization |
Styrene monomer |
|
Hydrochemolytic™ Oil |
|||
|
|
Mixed/Contaminated Plastics |
Gasification |
Syngas |
|
Selectively breaks down compatible materials into reusable intermediates or feedstocks |
Categorizing Advanced Plastic Recycling Types According to the Nova Institute
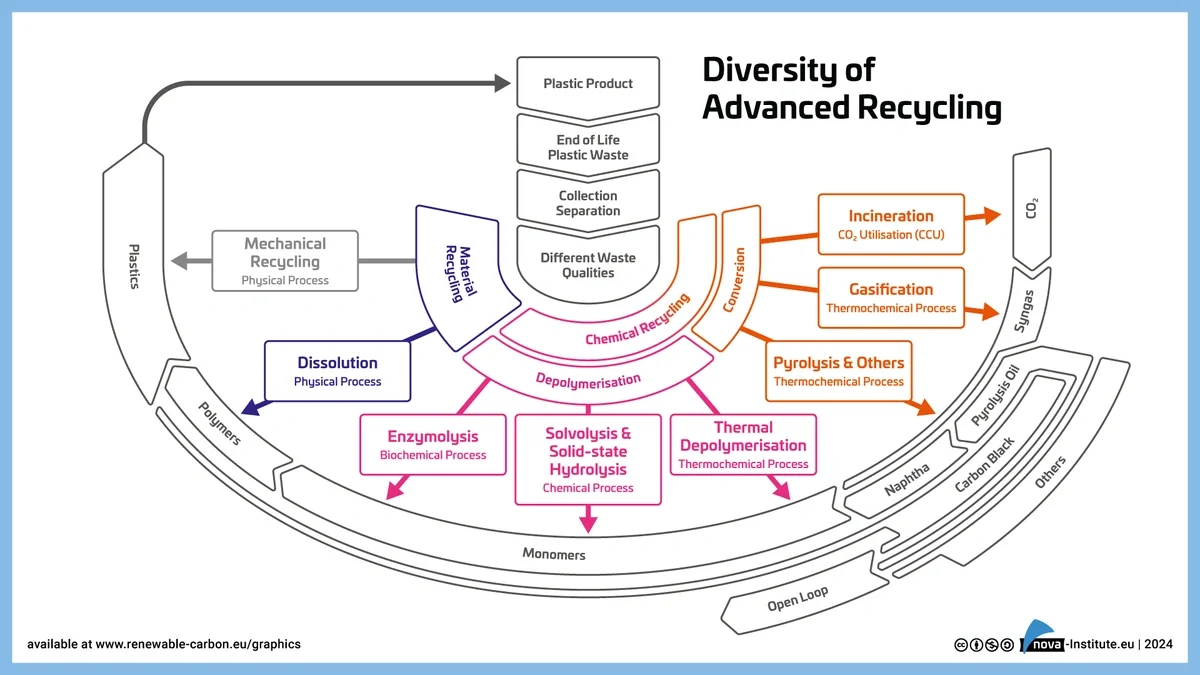
The Nova Institute offers a straightforward way to categorize advanced plastic recycling methods—which includes chemical and mechanical recycling. This framework helps simplify a complex landscape by focusing on how plastics are chemically or physically processed.
With this framework in mind, let’s explore each category in more detail, explaining how these methods work and which recycling technologies fall under each.
DepolymerizationBreaks plastic polymers back down into their original monomers using heat, pressure, or catalysts. Solvolysis Enzymolysis / Enzymatic Recycling Thermochemical Processes (such as Hydrogenolysis and Hydrocracking) |
ConversionTransforms plastics into entirely new chemical substances like fuels or syngas through high-temperature processes. Pyrolysis Gasification |
Dissolution / Solvent-Based PurificationNot considered true chemical recycling, dissolution cleans plastics by dissolving them in solvents to remove additives and impurities without breaking polymer chains. The extracted polymer can be processed with new additives to make new plastics. It is also used as a pre-treatment step to improve the efficiency of subsequent chemical recycling processes. |
What’s missing in this framework? How Hydrochemolytic™ Technology advances sustainable plastic recycling
Hydrochemolytic™ Technology recycling doesn’t cleanly fit into the Nova Institute’s established categories—it stands apart. Unlike established methods that rely on extreme heat, pressure, or pure solvents, this water-based process operates at lower temperatures with the help of custom catalysts. It doesn’t fall into either depolymerization, conversion, or dissolution, instead it draws elements from each. Aduro’s unique recycling mechanism works with water, and may signal the emergence of a new class of chemical recycling: one designed for precision, versatility, and a broader range of plastics, and potentially expanding the boundaries of what’s considered chemically recyclable.
Aduro’s process produces Hydrochemolytic™ Oil, which was recently shown to run as produced in pilot-scale steam-cracking trials, with no hydrotreatment or dilution. The successful testing demonstrated stable furnace operation and olefin yields comparable to conventional fossil feedstocks, marking an important validation milestone and highlighting its potential as a true drop-in circular feedstock for existing cracker infrastructure.
Categorizing chemical recycling types by mechanism: understanding how sustainable plastic recycling solutions break down plastics
Chemical recycling processes break plastics down using different mechanisms. These can include applying heat, using solvents, adding catalysts, or even introducing enzymes. Understanding these categories can help clarify why certain methods are better suited for specific plastic types or waste conditions.
Thermal-only methodsUse heat alone (without catalysts) to break down plastics.
|
Oxidative Thermal MethodsUse high heat and limited oxygen to convert plastics into syngas or other base chemicals.
|
Thermal + Catalytic MethodsUse high heat and catalysts together to cleave polymer chains more selectively or efficiently.
|
Solvent-Based MethodsUse solvents to dissolve plastics or chemically react with them without extreme heat.
|
Biological MethodsUse engineered enzymes to digest plastics under mild conditions.
|
Water-Catalyzed Hybrid MethodsUse unique combinations of water, moderate temperatures, and custom tailored catalysts to selectively break down plastics into high-value outputs—without relying on extreme heat or aggressive solvents. |
*Both Biological and Water-Catalyzed Hybrid methods are considered emerging or early-stage technologies., while Hydrochemolytic™ Technology is currently in pilot deployment with active development toward commercialization
Categorizing chemical recycling types by temperature and energy use: how plastic waste recycling methods differ
Temperature and energy demands vary widely between chemical recycling methods. Some rely on sustained high heat and pressure to break down plastics into fuels or gases, while others use solvents, catalysts, or even enzymes to operate at much lower energy levels. Understanding these differences can help identify which technologies are more energy-efficient, scalable, or suitable for specific waste streams.
High-Temperature, Energy-Intensive Methods |
Mid-Temperature, Catalyst-Driven Methods |
|
Pyrolysis and gasification require sustained high heat to break down plastic into fuels or syngas. Hydrocracking and hydrogenolysis also rely on heat and pressure, typically alongside hydrogen and specialized catalysts. These methods often demand substantial infrastructure and energy to maintain the required conditions. |
Depolymerization and solvolysis often use heat, but can operate at lower temperatures when aided by solvents or chemical catalysts. These methods can target specific polymers like PET or PS and typically aim to recover monomers for reuse. Aduro’s Hydrochemolytic™ technology uses water and catalysts at relatively low temperatures to transform plastics into usable components, offering an energy-efficient and potentially cleaner alternative to thermal methods.. |
Low-Temperature, Lower-Energy Methods |
|
|
Dissolution / Solvent-Based Purification uses room-temperature or gently heated solvents to separate pure plastic from additives or contaminants, without breaking the polymer chains. Enzymolysis / Enzymatic Recycling shows promise as a low-energy, biologically driven process using engineered enzymes to break down plastics. |
|
Closing thoughts on advanced plastic waste recycling, chemical recycling methods, and sustainable plastic solutions
As the demand for sustainable plastic solutions grows, chemical recycling is an important addition to the traditional recycling technologies to build a circular economy—one in which plastics are continuously repurposed rather than discarded. These technologies offer scalable solutions to manage plastic pollution by recovering value from materials that would otherwise be lost, and they expand what’s possible in the realm of plastic waste recycling.
With Extended Producer Responsibility (EPR) policies gaining traction, the pressure is on for producers to support smarter, more effective recycling systems. Understanding the range of chemical recycling technologies—and how they differ—helps all of us make better decisions, whether we're shaping policy, designing packaging, or just trying to stay informed.
We hope this guide has added clarity, made it easier to navigate the categories, and chemistry, and helped make these complex technologies a little easier to understand.